How to Verify the Accuracy of a pH Meter
Scope of application
1.1 This method is applicable for the determination of pH values in drinking water, surface water, and industrial wastewater.
1.2 The color, turbidity, colloidal substances, oxidants, reducing agents, and high salt content of water do not interfere with the measurement; However, in strongly acidic solutions with pH<1, there is a so-called "acid error" that can be measured based on acidity; in alkaline solutions with pH>10, due to the presence of a large number of sodium ions, errors occur, resulting in low readings, commonly known as "sodium difference". To eliminate the "sodium difference", in addition to using a specially designed "low sodium difference" electrode, a standard buffer solution similar to the pH value of the measured solution can also be used to calibrate the instrument.
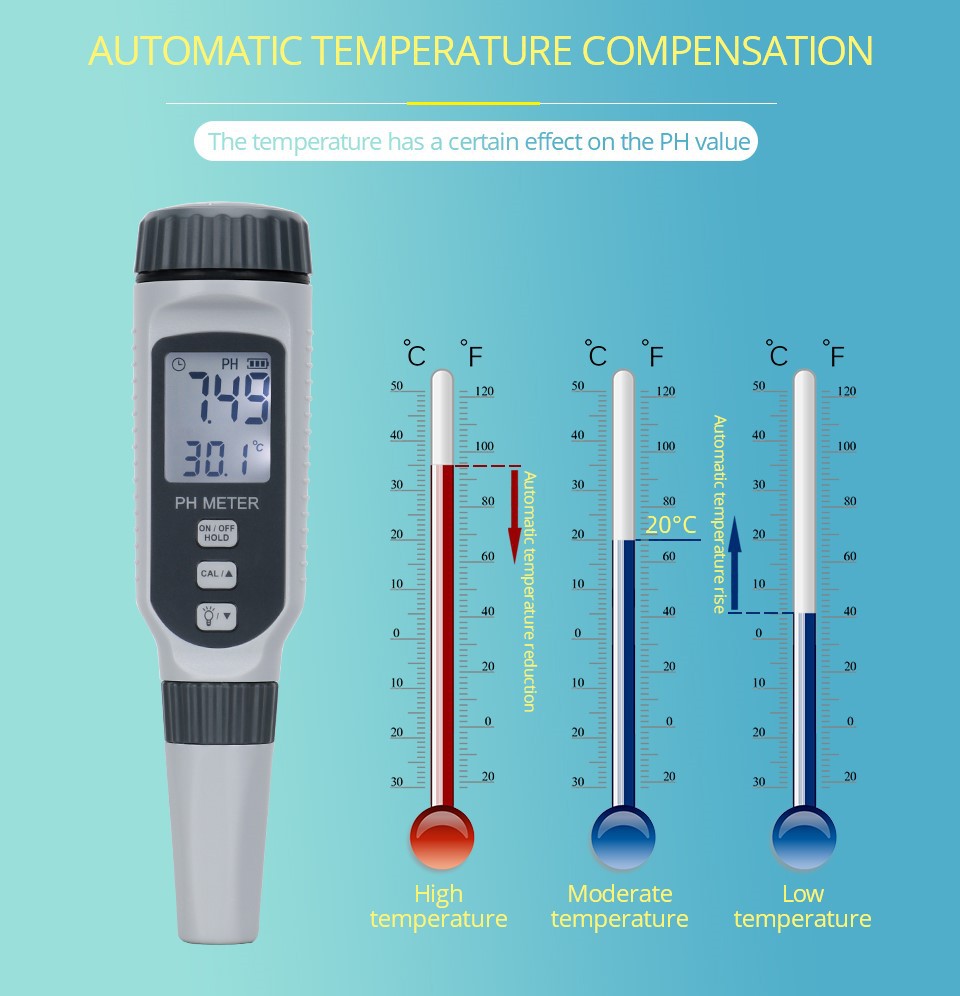
Temperature affects the potential of electrodes and the ionization equilibrium of water. Attention should be paid to adjusting the compensation device of the instrument to be consistent with the temperature of the solution, and ensuring that the temperature error between the measured sample and the standard buffer solution used for calibration instrument is within ± 1 ℃.
definition
PH is defined operationally. For solution x, measure the electromotive force EX of the reference electrode 1KC1 concentrated solution H solution X | H2 | pt of the galvanic cell. Replace the solution X with a standard pH solution S and measure the electromotive force ES of the cell
pH(X)=pH(S)+(Es-Ex)F/(RTIn10)
Therefore, the defined pH is a dimensionless quantity. PH has no theoretical significance and is defined as a practical definition. However, in the limited range of dilute aqueous solutions with a concentration of less than 0.1mo1.dm-3, which are neither strongly acidic nor strongly alkaline (Z
pH=-Log10[C(H+)y/(mo1.dm3)]±0.02
In the formula, C ((H+)) represents the concentration of hydrogen ion H+, and y represents the activity coefficient of a typical 1, -1 valent electrolyte in the solution.
principle
The pH value is obtained by measuring the electromotive force of the battery. This battery typically consists of a saturated calomel electrode as the reference electrode and a glass electrode as the indicator electrode. At 25 ℃, for every 1 pH unit change in the solution, the potential difference changes to 59.16 millivolts, which can be directly expressed as pH readings on the instrument. The temperature difference is compensated by a device on the instrument.