Application of pH Meters in Soil Acidity-Alkalinity Measurement
Summary of pH Measurement Methods
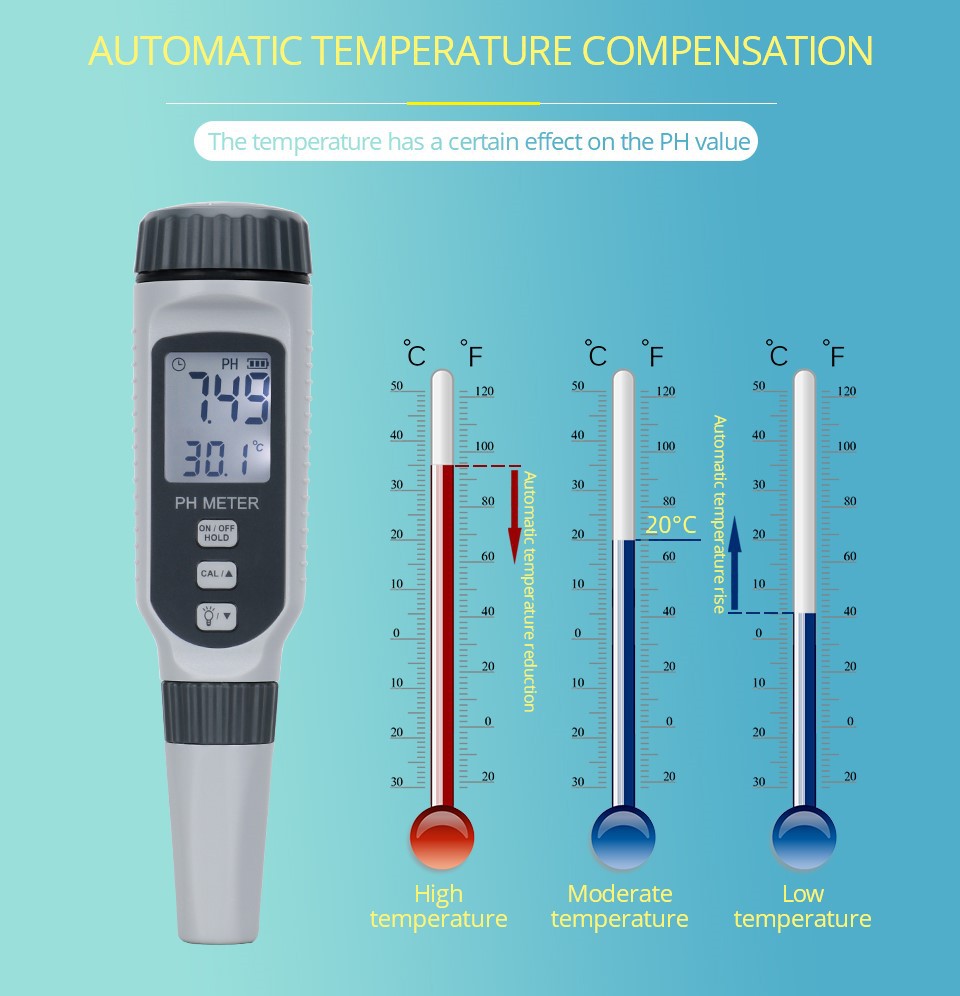
The determination of soil pH using the potentiometric method involves inserting pH glass electrodes and calomel electrodes into soil suspension or leachate, measuring their electromotive force values, and then converting them into pH values (acidity or alkalinity). The pH value can be directly read after being determined by a standard solution on an acidity meter. The water soil ratio has a significant impact on pH value, especially on the dilution effect of calcareous soil. It is advisable to adopt a small water soil ratio and set the soil pH at 1:1. At the same time, in addition to measuring the pH value of water soaked soil, the pH value of salt soaked soil should also be measured for acidic soil, that is, the soil H+should be leached with 1mol/L potassium chloride solution and measured by potentiometry.
PH meter measuring reagent
① 1mol/L potassium chloride solution: Weigh 74.6 ɡ potassium chloride (chemically pure) and dissolve it in 800ml of water. Adjust the batch pH of the solution to 5.5-6.0 with dilute potassium hydroxide and dilute hydrochloric acid, and dilute to 1L;
② PH 4.01 (25 ℃) standard buffer solution: Weigh 10.21 potassium hydrogen phthalate, which has been dried at 110-120 ℃ for 2-3 hours, and
dissolve it in water. Transfer it into a 1L volumetric flask, dilute with water, and store it in a polyethylene flask;
③ Ph6.87 (25 ℃) standard buffer solution: Weigh 3.533g of disodium hydrogen phosphate and 3.388g of potassium dihydrogen phosphate, which have been dried at 110-130 ℃ for 2-3 hours, and dissolve them in water. Transfer them into a 1L volumetric flask, make up to volume with water, and store them in a polyethylene flask;
④ Ph9.18 (25 ℃) standard buffer solution: Weigh 3.800g of borax (Na2B4O7 · 10H2O) that has been equilibrated and dissolve it in CO2 free water. Transfer it into a 1L volumetric flask, make up to volume with water, and store it in a polyethylene flask;