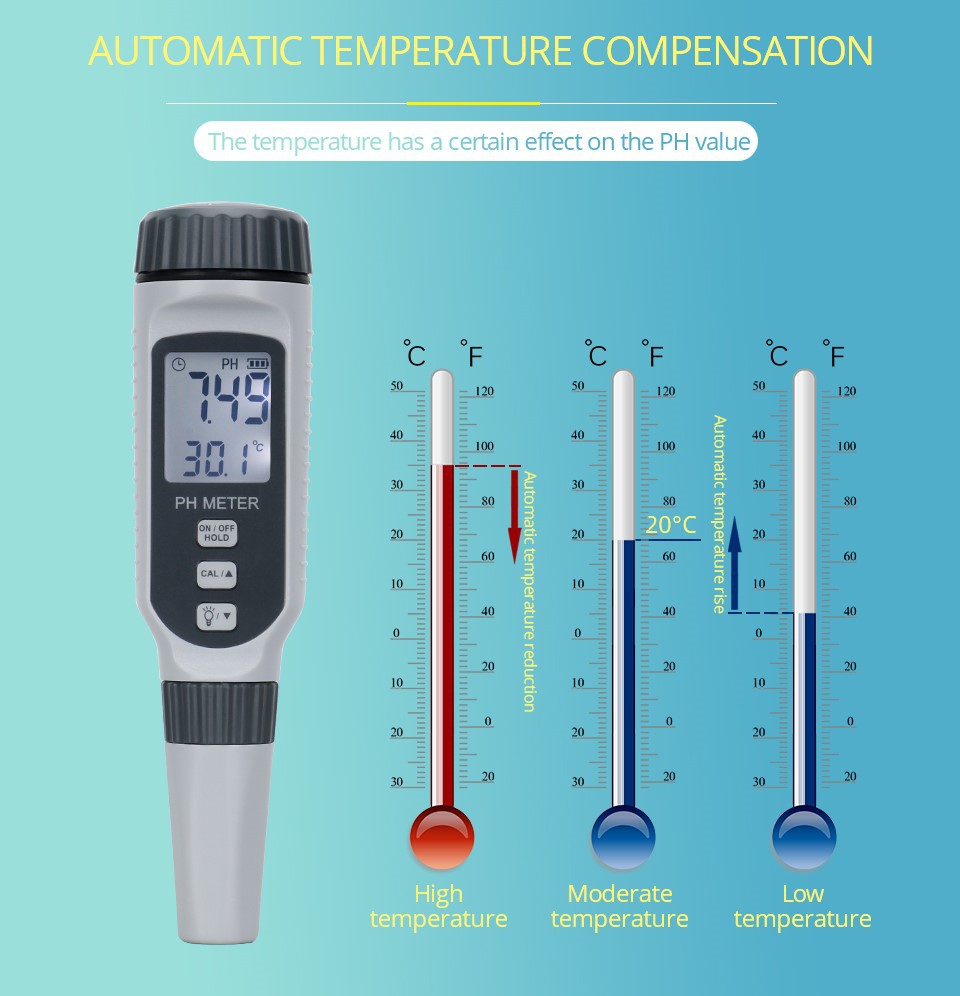
Difference Between Acidimeter and pH Meter
The acidity meter and pH meter are the same, and are high-precision industrial online automatic continuous detection of acidity and alkalinity analytical instruments.
The pH meter, abbreviated as pH meter, consists of two parts: electrode and ammeter. It is a commonly used instrument and equipment, consisting of a reference electrode, a glass electrode, and an ammeter. Widely used in industries, agriculture, scientific research, environmental protection, and other fields.
Acidimeter is a commonly used instrument and equipment, also known as pH meter. The BPH-200A series pH meter is mainly used for precise measurement of the pH value of liquid media, and can also measure the MV value of ion electrode potential when equipped with corresponding ion selective electrodes. It is widely used in industries, agriculture, scientific research, environmental protection and other fields. This instrument is also an essential inspection equipment for QS and HACCP certification in food factories and drinking water plants.
An acidimeter is an instrument used to measure the pH value of a solution. The main body of an acidity meter is a precision potentiometer. When measuring, the composite electrode is inserted into the measured solution, and different electromotive forces are generated due to the acidity (hydrogen ion concentration) of the measured solution. It is amplified by a DC amplifier and finally indicated by a reading indicator (voltmeter) to determine the pH value of the measured solution. The acidity meter can be used within the pH range of 0-14.
Instructions for using a laboratory pH meter
(1) When using for the first time, due to the relatively dry electrode probe, it will affect the measurement accuracy of the instrument. Therefore, soak the electrode in a 3mol/L potassium chloride solution for 2 hours first.
(2) Preparation of 3mol/L potassium chloride solution: Weigh 223.65g of potassium chloride and dissolve it in one liter of distilled or deionized water. Dissolve the reagent completely to obtain a 3mol/L potassium chloride solution.
(3) Replace the electrode once a year (regardless of whether the electrode is functional or not).